 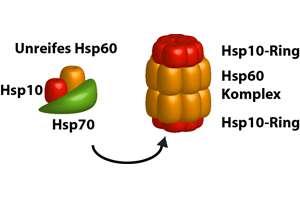
Aided by a set of smaller hsp40 proteins (not shown), ATP-bound hsp70 molecules grasp their target protein and then hydrolyze ATP to ADP, undergoing conformational changes that cause the hsp70 molecules to associate even more tightly with the target. These proteins act early, recognizing a small stretch of hydrophobic amino acids on a protein’s surface. The hsp70 family of molecular chaperones. The hsp70 machinery acts early in the life of many proteins (often before the protein leaves the ribosome), with each monomer of hsp70 binding to a string of about four or five hydrophobic amino acids ( see figure below ) In other respects, the two types of hsp proteins function differently. These hsps share an affinity for the exposed hydrophobic patches on incompletely folded proteins, and they hydrolyze ATP, often binding and releasing their protein substrate with each cycle of ATP hydrolysis. The hsp60 and hsp70 proteins each work with their own small set of associated proteins when they help other proteins to fold. Thus, mitochondria contain their own hsp60 and hsp70 molecules that are distinct from those that function in the cytosol and a special hsp70 (called BIP) helps to fold proteins in the endoplasmic reticulum. Different members of these families function in different organelles. There are several major families of molecular chaperones, including the hsp60 and hsp70 proteins. This reflects the operation of a feedback system that responds to an increase in misfolded proteins (such as those produced by elevated temperatures) by boosting the synthesis of the chaperones that help these proteins refold. Many molecular chaperones are called heat-shock proteins (designated hsp), because they are synthesized in dramatically increased amounts after a brief exposure of cells to an elevated temperature (for example, 42☌ for cells that normally live at 37☌). There are several types of chaperones once bound to an incorrectly folded protein, they ultimately release it in a way that gives the protein another chance to fold correctly.Ĭells Utilize Several Types of Chaperones Chaperones prevent this from happening in normal proteins by binding to the exposed hydrophobic surfaces using hydrophobic surfaces of their own. In some cases of inherited human diseases, aggregates do form and can cause severe symptoms and even death. The binding of these exposed hydrophobic surfaces to each other is what causes off-pathway conformations to irreversibly aggregate. Molecular chaperones specifically recognize incorrect, off-pathway configurations by their exposure of hydrophobic surfaces, which in correctly folded proteins are typically buried in the interior. Some of these off-pathway configurations would aggregate and be left as irreversible dead ends of nonfunctional (and potentially dangerous) structures. Without chaperones, some of these pathways would not lead to the correctly folded (and most stable) form because the protein would become “kinetically trapped” in structures that are off-pathway. Molecular chaperones are useful for cells because there are many different folding paths available to an unfolded or partially folded protein. 
Most proteins probably do not fold correctly during their synthesis and require a special class of proteins called molecular chaperones to do so. Machines, that help other machines to assemble properly, are by themselves subject to errors, and life has inbuilt mechanisms to fix as well these machines that help fixing other machines !! And it has been shown that GroES, a co-chaperone assists in folding GroEL. Now, amazingly, even these very own proteins, named GroEL, which help misfolded proteins to fold properly, can also misfold. They have been shown to interact with up to 30% of the cell’s proteins, so their importance is real. Spontaneous folding is quite rapid (milliseconds to seconds) for many proteins, but many large, critical proteins fail to find by themselves the right shape and, without help, would become only so much molecular waste.So when a protein misfolds, other proteins, named chaperonins, help proteins fold into the right shape. If the first proteins fell into these death valleys, life on Earth would never have appeared. If it folds into the wrong shape, a protein is useless. Properly folded proteins are essential for life because they conduct most of the necessary functions in a cell. Proteins,marvelous pieces of chemical nanoengineering, in order to become functional, must fold from linear, to specific 3-D forms. Evidently, such a process/system requires the highest sort of engineering artistry and foreknowledge of what could be wrong and how to fix the errors. 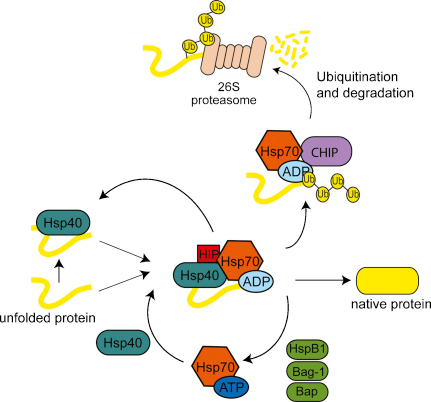
All fully set up to operate in an independent, fully automated, robot-like manner. Imagine machines, that fix machines, which help to fix other machines. Molecular Chaperones Help Guide the Folding of Most Proteins 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
